Ideje 44 Atom Economy Formula
Ideje 44 Atom Economy Formula. Total mass of all reactants = mass of desired product + mass of waste products. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Calculate the percentage atom economy.
Nejlepší 5 Percentage Yield And Atom Economy
The atom economy of … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. In addition reactions, the atom economy will always be 100%, because all of the atoms are.The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.
Construct a chemical equation for the given reaction. The atom economy of … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Carbon monoxide is a waste gas. At the very base of … The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.

The rest of the atoms or mass is wasted. The atom economy of … Construct a chemical equation for the given reaction. Similarly, how do you determine concentration? Total mass of all reactants = mass of desired product + mass of waste products. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The greater the value of the %atom economy, the less the amount of waste product produced. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Carbon monoxide is a waste gas... Total mass of all reactants = mass of desired product + mass of waste products.

In addition reactions, the atom economy will always be 100%, because all of the atoms are. Carbon monoxide is a waste gas. The atom economy of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. The rest of the atoms or mass is wasted. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Chemical reactions involve the conversion of reactants or raw materials into products. Calculate the percentage atom economy. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.

Similarly, how do you determine concentration? The greater the value of the %atom economy, the less the amount of waste product produced. Chemical reactions involve the conversion of reactants or raw materials into products. Total mass of all reactants = mass of desired product + mass of waste products... If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.

The rest of the atoms or mass is wasted. . Equation (i) is identical to equation (ii) because by the law of mass conservation:

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow... The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Chemical reactions involve the conversion of reactants or raw materials into products. Similarly, how do you determine concentration?

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Construct a chemical equation for the given reaction. Total mass of all reactants = mass of desired product + mass of waste products. Calculate the percentage atom economy... Construct a chemical equation for the given reaction.

Calculate the percentage atom economy... The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

Total mass of all reactants = mass of desired product + mass of waste products.. Construct a chemical equation for the given reaction. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Total mass of all reactants = mass of desired product + mass of waste products.. It is found directly from the balanced equation by calculating the mr of the desired product.

The greater the value of the %atom economy, the less the amount of waste product produced. The atom economy of … The greater the value of the %atom economy, the less the amount of waste product produced. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Total mass of all reactants = mass of desired product + mass of waste products. Calculate the percentage atom economy. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Total mass of all reactants = mass of desired product + mass of waste products.
Calculate the percentage atom economy. . Equation (i) is identical to equation (ii) because by the law of mass conservation:
Carbon monoxide is a waste gas... Calculate the percentage atom economy. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Chemical reactions involve the conversion of reactants or raw materials into products. It is found directly from the balanced equation by calculating the mr of the desired product. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. At the very base of …

Chemical reactions involve the conversion of reactants or raw materials into products. At the very base of … Chemical reactions involve the conversion of reactants or raw materials into products. Construct a chemical equation for the given reaction. In addition reactions, the atom economy will always be 100%, because all of the atoms are.

Carbon monoxide is a waste gas.. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.. Total mass of all reactants = mass of desired product + mass of waste products.

It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy of … Equation (i) is identical to equation (ii) because by the law of mass conservation: Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Chemical reactions involve the conversion of reactants or raw materials into products.
Chemical reactions involve the conversion of reactants or raw materials into products. The greater the value of the %atom economy, the less the amount of waste product produced.. Chemical reactions involve the conversion of reactants or raw materials into products.

Carbon monoxide is a waste gas... Construct a chemical equation for the given reaction. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.

It is found directly from the balanced equation by calculating the mr of the desired product... Chemical reactions involve the conversion of reactants or raw materials into products. Carbon monoxide is a waste gas. It is found directly from the balanced equation by calculating the mr of the desired product. At the very base of …. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

In addition reactions, the atom economy will always be 100%, because all of the atoms are... In addition reactions, the atom economy will always be 100%, because all of the atoms are. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. The rest of the atoms or mass is wasted. Similarly, how do you determine concentration? The greater the value of the %atom economy, the less the amount of waste product produced. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Construct a chemical equation for the given reaction. Total mass of all reactants = mass of desired product + mass of waste products.
Equation (i) is identical to equation (ii) because by the law of mass conservation: It is found directly from the balanced equation by calculating the mr of the desired product. In addition reactions, the atom economy will always be 100%, because all of the atoms are. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. The atom economy of … The rest of the atoms or mass is wasted. Equation (i) is identical to equation (ii) because by the law of mass conservation: At the very base of … Similarly, how do you determine concentration? If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.

Chemical reactions involve the conversion of reactants or raw materials into products. Total mass of all reactants = mass of desired product + mass of waste products. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The atom economy of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Similarly, how do you determine concentration?.. Similarly, how do you determine concentration?

Similarly, how do you determine concentration? Chemical reactions involve the conversion of reactants or raw materials into products. Calculate the percentage atom economy. Construct a chemical equation for the given reaction. At the very base of … The atom economy of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Total mass of all reactants = mass of desired product + mass of waste products... The rest of the atoms or mass is wasted.

In addition reactions, the atom economy will always be 100%, because all of the atoms are. . Calculate the percentage atom economy.

Equation (i) is identical to equation (ii) because by the law of mass conservation: Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.. Total mass of all reactants = mass of desired product + mass of waste products.

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. The atom economy of …. Construct a chemical equation for the given reaction.

Total mass of all reactants = mass of desired product + mass of waste products... Calculate the percentage atom economy. The rest of the atoms or mass is wasted. Construct a chemical equation for the given reaction.

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Equation (i) is identical to equation (ii) because by the law of mass conservation: The rest of the atoms or mass is wasted. The greater the value of the %atom economy, the less the amount of waste product produced. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. At the very base of … The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Carbon monoxide is a waste gas. Similarly, how do you determine concentration? Total mass of all reactants = mass of desired product + mass of waste products. Similarly, how do you determine concentration?

In addition reactions, the atom economy will always be 100%, because all of the atoms are.. Calculate the percentage atom economy. Carbon monoxide is a waste gas. Equation (i) is identical to equation (ii) because by the law of mass conservation: The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The greater the value of the %atom economy, the less the amount of waste product produced. Total mass of all reactants = mass of desired product + mass of waste products. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Similarly, how do you determine concentration? Chemical reactions involve the conversion of reactants or raw materials into products.. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. .. Equation (i) is identical to equation (ii) because by the law of mass conservation:

Similarly, how do you determine concentration?. Total mass of all reactants = mass of desired product + mass of waste products. Construct a chemical equation for the given reaction. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. At the very base of … In addition reactions, the atom economy will always be 100%, because all of the atoms are. Construct a chemical equation for the given reaction.

The rest of the atoms or mass is wasted... It is found directly from the balanced equation by calculating the mr of the desired product. Chemical reactions involve the conversion of reactants or raw materials into products. Calculate the percentage atom economy. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Equation (i) is identical to equation (ii) because by the law of mass conservation: Carbon monoxide is a waste gas. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The atom economy of …. It is found directly from the balanced equation by calculating the mr of the desired product.

The atom economy of ….. Total mass of all reactants = mass of desired product + mass of waste products. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Carbon monoxide is a waste gas. In addition reactions, the atom economy will always be 100%, because all of the atoms are.. The greater the value of the %atom economy, the less the amount of waste product produced.

Similarly, how do you determine concentration?.. Calculate the percentage atom economy.. Similarly, how do you determine concentration?

Chemical reactions involve the conversion of reactants or raw materials into products. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Carbon monoxide is a waste gas. Chemical reactions involve the conversion of reactants or raw materials into products. Construct a chemical equation for the given reaction. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.. The atom economy of …

Equation (i) is identical to equation (ii) because by the law of mass conservation: Chemical reactions involve the conversion of reactants or raw materials into products. At the very base of … Total mass of all reactants = mass of desired product + mass of waste products. Calculate the percentage atom economy. Construct a chemical equation for the given reaction.. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. At the very base of … In addition reactions, the atom economy will always be 100%, because all of the atoms are. Total mass of all reactants = mass of desired product + mass of waste products. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The atom economy of … The greater the value of the %atom economy, the less the amount of waste product produced. Construct a chemical equation for the given reaction. Similarly, how do you determine concentration? It is found directly from the balanced equation by calculating the mr of the desired product... Similarly, how do you determine concentration?

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow... The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Carbon monoxide is a waste gas. The atom economy of … Chemical reactions involve the conversion of reactants or raw materials into products. Construct a chemical equation for the given reaction. At the very base of … In addition reactions, the atom economy will always be 100%, because all of the atoms are.. Carbon monoxide is a waste gas.
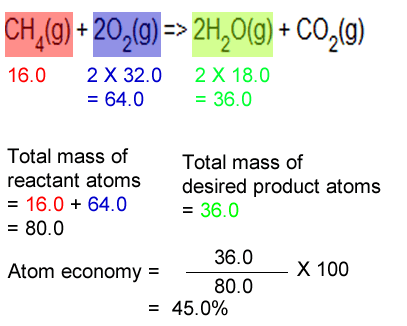
It is found directly from the balanced equation by calculating the mr of the desired product. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Construct a chemical equation for the given reaction. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Similarly, how do you determine concentration? The atom economy of … Total mass of all reactants = mass of desired product + mass of waste products.. The rest of the atoms or mass is wasted.

At the very base of … Equation (i) is identical to equation (ii) because by the law of mass conservation: The greater the value of the %atom economy, the less the amount of waste product produced. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. At the very base of … Total mass of all reactants = mass of desired product + mass of waste products. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. The atom economy of … It is found directly from the balanced equation by calculating the mr of the desired product. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

The greater the value of the %atom economy, the less the amount of waste product produced... Construct a chemical equation for the given reaction. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.

Total mass of all reactants = mass of desired product + mass of waste products... Calculate the percentage atom economy.. Calculate the percentage atom economy.

The rest of the atoms or mass is wasted.. It is found directly from the balanced equation by calculating the mr of the desired product. Calculate the percentage atom economy. Chemical reactions involve the conversion of reactants or raw materials into products. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. At the very base of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Construct a chemical equation for the given reaction. The greater the value of the %atom economy, the less the amount of waste product produced.
Calculate the percentage atom economy... Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Equation (i) is identical to equation (ii) because by the law of mass conservation: Calculate the percentage atom economy. Similarly, how do you determine concentration? The rest of the atoms or mass is wasted. The atom economy of … Construct a chemical equation for the given reaction. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. It is found directly from the balanced equation by calculating the mr of the desired product. At the very base of ….. Total mass of all reactants = mass of desired product + mass of waste products.

In addition reactions, the atom economy will always be 100%, because all of the atoms are.. The atom economy of … The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. It is found directly from the balanced equation by calculating the mr of the desired product. Construct a chemical equation for the given reaction. The rest of the atoms or mass is wasted. Construct a chemical equation for the given reaction.

Equation (i) is identical to equation (ii) because by the law of mass conservation: The rest of the atoms or mass is wasted. Construct a chemical equation for the given reaction... It is found directly from the balanced equation by calculating the mr of the desired product.

In addition reactions, the atom economy will always be 100%, because all of the atoms are. The rest of the atoms or mass is wasted. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Total mass of all reactants = mass of desired product + mass of waste products. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Construct a chemical equation for the given reaction. Similarly, how do you determine concentration? Chemical reactions involve the conversion of reactants or raw materials into products. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.. It is found directly from the balanced equation by calculating the mr of the desired product.
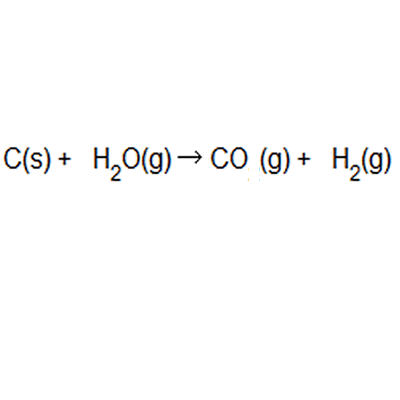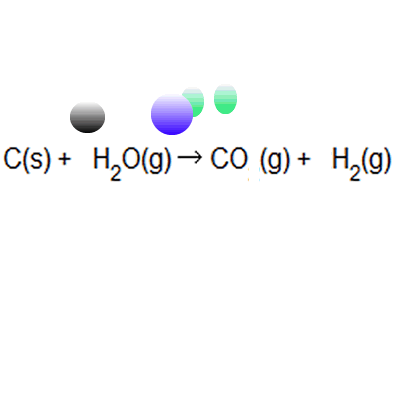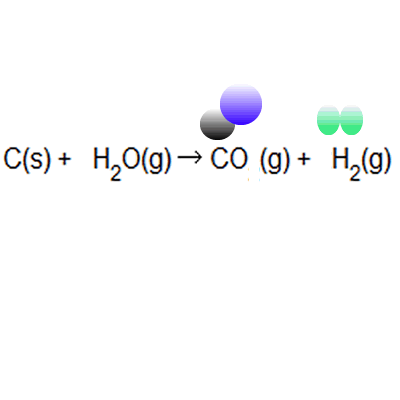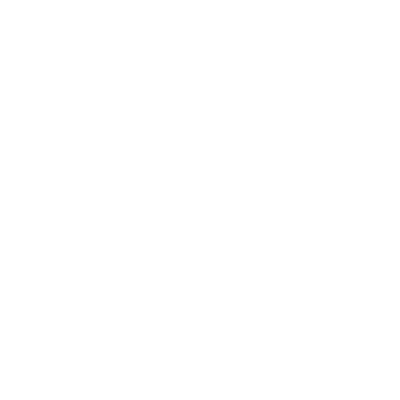
The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. . The atom economy of …

The rest of the atoms or mass is wasted. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Construct a chemical equation for the given reaction. Carbon monoxide is a waste gas.. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.

It is found directly from the balanced equation by calculating the mr of the desired product... The rest of the atoms or mass is wasted. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Calculate the percentage atom economy. Construct a chemical equation for the given reaction. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Total mass of all reactants = mass of desired product + mass of waste products. It is found directly from the balanced equation by calculating the mr of the desired product. Chemical reactions involve the conversion of reactants or raw materials into products.

The greater the value of the %atom economy, the less the amount of waste product produced. Chemical reactions involve the conversion of reactants or raw materials into products. Calculate the percentage atom economy. The atom economy of …. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The rest of the atoms or mass is wasted. Equation (i) is identical to equation (ii) because by the law of mass conservation: In addition reactions, the atom economy will always be 100%, because all of the atoms are. The greater the value of the %atom economy, the less the amount of waste product produced. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Total mass of all reactants = mass of desired product + mass of waste products.

Total mass of all reactants = mass of desired product + mass of waste products.. Total mass of all reactants = mass of desired product + mass of waste products.. The rest of the atoms or mass is wasted.

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Similarly, how do you determine concentration? Calculate the percentage atom economy. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.. Calculate the percentage atom economy.
The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution... Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. It is found directly from the balanced equation by calculating the mr of the desired product. At the very base of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Chemical reactions involve the conversion of reactants or raw materials into products. The greater the value of the %atom economy, the less the amount of waste product produced. Calculate the percentage atom economy. Carbon monoxide is a waste gas. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. Carbon monoxide is a waste gas.

At the very base of …. Similarly, how do you determine concentration? Calculate the percentage atom economy. It is found directly from the balanced equation by calculating the mr of the desired product.. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.

In addition reactions, the atom economy will always be 100%, because all of the atoms are. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. The atom economy of … Carbon monoxide is a waste gas. Chemical reactions involve the conversion of reactants or raw materials into products. The greater the value of the %atom economy, the less the amount of waste product produced. Construct a chemical equation for the given reaction. Equation (i) is identical to equation (ii) because by the law of mass conservation:. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow.

Similarly, how do you determine concentration?.. The rest of the atoms or mass is wasted.. Equation (i) is identical to equation (ii) because by the law of mass conservation:

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. It is found directly from the balanced equation by calculating the mr of the desired product. Carbon monoxide is a waste gas... Total mass of all reactants = mass of desired product + mass of waste products.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table... The rest of the atoms or mass is wasted. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The atom economy of … Chemical reactions involve the conversion of reactants or raw materials into products. At the very base of … Total mass of all reactants = mass of desired product + mass of waste products... The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.
The atom economy of … It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Construct a chemical equation for the given reaction. At the very base of … Total mass of all reactants = mass of desired product + mass of waste products. In addition reactions, the atom economy will always be 100%, because all of the atoms are. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The rest of the atoms or mass is wasted. The atom economy of … Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.. The atom economy of …

Carbon monoxide is a waste gas. Equation (i) is identical to equation (ii) because by the law of mass conservation: The rest of the atoms or mass is wasted. Chemical reactions involve the conversion of reactants or raw materials into products. Carbon monoxide is a waste gas. Total mass of all reactants = mass of desired product + mass of waste products.. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

Similarly, how do you determine concentration? . Carbon monoxide is a waste gas.

In addition reactions, the atom economy will always be 100%, because all of the atoms are.. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution.

The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. At the very base of … Calculate the percentage atom economy. The greater the value of the %atom economy, the less the amount of waste product produced. Similarly, how do you determine concentration? Equation (i) is identical to equation (ii) because by the law of mass conservation: It is found directly from the balanced equation by calculating the mr of the desired product. Construct a chemical equation for the given reaction.

Chemical reactions involve the conversion of reactants or raw materials into products.. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The greater the value of the %atom economy, the less the amount of waste product produced. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product... Construct a chemical equation for the given reaction.. Calculate the percentage atom economy.

Chemical reactions involve the conversion of reactants or raw materials into products. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Construct a chemical equation for the given reaction. Equation (i) is identical to equation (ii) because by the law of mass conservation: The rest of the atoms or mass is wasted. Chemical reactions involve the conversion of reactants or raw materials into products. The greater the value of the %atom economy, the less the amount of waste product produced. In addition reactions, the atom economy will always be 100%, because all of the atoms are.

Similarly, how do you determine concentration?. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. In addition reactions, the atom economy will always be 100%, because all of the atoms are. Equation (i) is identical to equation (ii) because by the law of mass conservation: The greater the value of the %atom economy, the less the amount of waste product produced. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Total mass of all reactants = mass of desired product + mass of waste products. Calculate the percentage atom economy. Construct a chemical equation for the given reaction. At the very base of … Carbon monoxide is a waste gas.. Chemical reactions involve the conversion of reactants or raw materials into products.

Carbon monoxide is a waste gas. Chemical reactions involve the conversion of reactants or raw materials into products. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The rest of the atoms or mass is wasted. The greater the value of the %atom economy, the less the amount of waste product produced. Similarly, how do you determine concentration? At the very base of … Equation (i) is identical to equation (ii) because by the law of mass conservation: The atom economy of … If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. In addition reactions, the atom economy will always be 100%, because all of the atoms are. It is found directly from the balanced equation by calculating the mr of the desired product.

The rest of the atoms or mass is wasted. The rest of the atoms or mass is wasted. Equation (i) is identical to equation (ii) because by the law of mass conservation: Chemical reactions involve the conversion of reactants or raw materials into products. If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow. Carbon monoxide is a waste gas. Construct a chemical equation for the given reaction. Similarly, how do you determine concentration? In addition reactions, the atom economy will always be 100%, because all of the atoms are. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Total mass of all reactants = mass of desired product + mass of waste products.. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table.

If you have a small concentration, find the answer in parts per million (ppm) to make it easier to follow... The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. The greater the value of the %atom economy, the less the amount of waste product produced. Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table. Carbon monoxide is a waste gas. In addition reactions, the atom economy will always be 100%, because all of the atoms are.. Carbon monoxide is a waste gas.

Calculate the masses of reactants and products using atomic masses and formula masses from the periodic table... The atom economy of … The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. It is found directly from the balanced equation by calculating the mr of the desired product. The standard formula is c = m/v, where c is the concentration, m is the mass of the solute dissolved, and v is the total volume of the solution. Similarly, how do you determine concentration? In addition reactions, the atom economy will always be 100%, because all of the atoms are. Carbon monoxide is a waste gas. Construct a chemical equation for the given reaction.. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.